Select Your Region
Medical Silicone Compliance: Why It Matters

Healthcare-grade silicone elastomers must meet strict standards for purity, clarity, durability, and biocompatibility. Momentive’s Silopren™ LSR 4000 series is engineered to comply with these requirements, making it ideal for both disposable and reusable medical devices.
Key Compliance Features

- Biocompatibility: Safe for contact with body fluids and medications
- Regulatory Standards: Meets stringent healthcare regulations and ISO, USP Class VI compliance, and FDA requirements
- Sterilization Resistance: Maintains performance after repeated autoclave, gamma, or EO sterilization
- Primerless Adhesion: Bonds to substrates like PC, PBT, and copolyesters without post-curing
- Aging and Chemical Stability: Withstands harsh environments and extreme temperatures
Medical Silicone Specification Checklist
When selecting silicone for healthcare applications, ensure your material meets the critical specifications below.Requirement
|
Silopren™ LSR 4000 Series Advantage
|
|---|---|
Biocompaibility
|
Compliant with ISO, Class VI, FDA standards
|
Sterilization Durability
|
Stable after multiple sterilization (autoclave, gamma, or EO) cycles
|
Transparency & Clarity
|
High optical clarity for diagnostic devices
|
Durometer Range
|
Wide Shore A hardness (~30A) options
|
Fast Cure & Tear Strength
|
Supports high-speed injection molding processes
|
Self-Bonding Capability
|
Primerless adhesion to plastics and metals
|
Design Flexibility
|
Enables soft-hard overmolding and insert molding
|
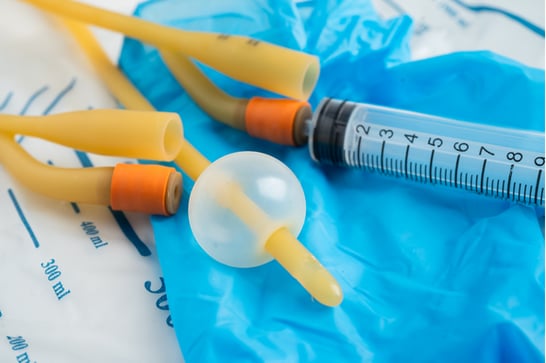
- General Medical Parts: O-rings, gaskets, seals, valves, diaphragms
- Drug Delivery Systems: Catheters, tubing, dosage control devices
- Respiratory & Surgical: Masks, valves, wound drains
- Dental & Orthopedic: Impression molds, prosthetic components
Download Medical Silicone Checklist
Help your end-customer material meets FDA, ISO, and USP Class VI requirements before it reaches qualification.
Or if you still require technical assistance, click the button below to talk to a silicone expert within seconds!
Click to download the compliance checklist
Or if you still require technical assistance, click the button below to talk to a silicone expert within seconds!
Are you in the correct region?
We’ve detected that you are located in a different region than the region selected on the website. Would you like to change your region?
Current Region: English - Canada