Select Your Region
Tyzor® - Non-Toxic Organometallic Catalysts

Esterification Catalysts
Esterification is a fundamental reaction in synthesizing esters, which are commercially important in paint and coating formulations, polymers, pharmaceuticals, and biodiesels. Classic esterification reactions between a carboxylic acid and alcohol, as in the case of Fischer esterification, typically require a strong acid catalyst to increase yield, reduce reaction times, and lower reaction temperature. Strong acids like sulfuric acid or sulfonic acid are often used as catalysts to improve synthesis rates and esterification reaction conditions. Organometallic chemistry can also be used as an esterification catalyst in place of an acid to produce esters. However, these catalysts often contain tin, antimony, or other metals. While effective, these traditional catalysts can be quite dangerous and have long-lasting environmental impacts.Tyzor®: Non-Toxic Catalysts
Tyzor® titanates are chemical compounds with organic functional groups linked to a central titanium atom via an ester bond. Tyzor® titanates are useful as nontoxic organometallic catalysts for esterification and transesterification reactions. In esterification reactions, Tyzor® acts as a metal-based Lewis acid and increases the reactivity of the substrate, resulting in faster synthesis and a higher yield. This makes Tyzor® an excellent choice for ester synthesis, improving reaction efficiency and reducing the hazards and environmental impact associated with using strong acids and organometallic catalysts containing tin or antimony. To highlight the usefulness of Tyzor® titanates as Lewis acid catalysts for esterification, Tyzor® TPT and Tyzor® TE were compared to sulfuric acid for catalysis of the direct esterification of adipic acid to bis-2-ethylhexyl adipate. The table and graph below show the reaction efficiency as a percent conversion over time.| Catalyst | Weight (%)
|
% Conversion 60 min (%)
|
in 75 min (%)
|
in 90 min (%)
|
in 105 min (%)
|
|---|---|---|---|---|---|
| Tyzor® TPT | 1.3
|
96
|
100
|
N/A
|
N/A
|
| Tyzor® TE | 1.3
|
78
|
90
|
96
|
100
|
| H2SO4 | 0.6
|
89
|
97
|
100
|
N/A
|
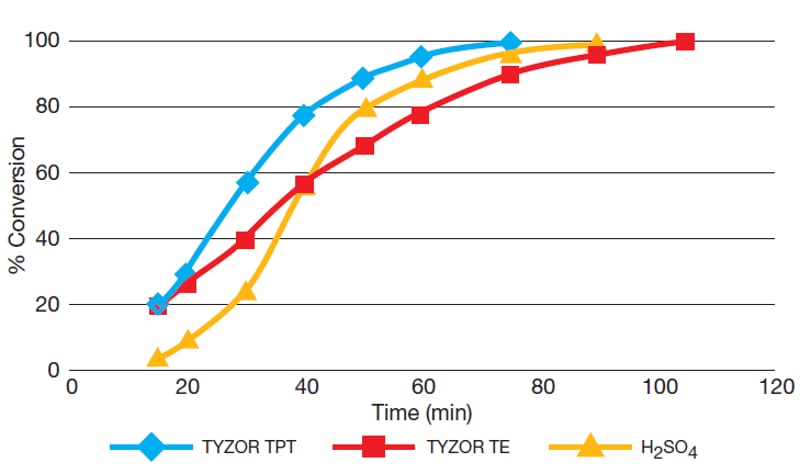
As seen in the data above, Tyzor® TPT is highly reactive in esterification reactions, achieving complete catalysis in 75 minutes. This shows that the Tyzor® TPT titanate provides a faster reaction time and more efficient catalysis than sulfuric acid. Although Tyzor® TPT is the most active chemistry in the Tyzor® product line and will react very quickly, it may not always be the most economical for esterification catalysis because its alkoxy titanate chemistry is highly sensitive to water, alcohols, and acids. If these are present or evolve during esterification, synthesis will not be as efficient. In these cases, titanium chelate chemistry, such as Tyzor® TE, will be more practical to use as a metal-based Lewis acid catalyst for esterification. Tyzor® TE first undergoes hydrolysis to form a reactive hydroxy titanium chelate, which is stable in water and catalyzes the esterification reaction.
How to Use Tyzor® for Esterification and Transesterification
Tyzor® Lewis acid catalysts are soluble in nonaqueous media and can be added to the reaction mixture at any point in the process. In the esterification of alcohols and carboxylic acids, as in the reaction mentioned above, it is recommended to add Tyzor® catalysts at a temperature above 150°C and sustain a reaction temperature between 180°C and 220°C to achieve the most efficient conversion. It is also particularly important to remove any water produced during esterification or transesterification as rapidly as possible to avoid deactivating the moisture-sensitive Tyzor® Lewis acid catalyst. In most cases, a polymerization inhibitor and nitrogen blanket are also useful if the corresponding ester being prepared is moisture sensitive. Catalyst concentration can vary based on the reaction, but typically, 0.1–0.5 wt% of Tyzor® based on the weight of the total product expected from the reaction is sufficient.Catalyst Removal
In some cases, it may be necessary to remove the Tyzor® catalyst from the ester product. While Tyzor® is generally soluble in product esters, it can easily be removed by conversion to its insoluble product of hydrolysis. This is accomplished by agitation with a small number of aqueous media followed by steam stripping, which will hydrolyze Tyzor® catalysts. The resulting chemicals are insoluble titanium oxides that can be removed by filtration.Summary
Tyzor® organic titanates and titanium chelates are safer Lewis acid catalysts for use in esterification and transesterification reactions to produce high-purity esters with higher efficiency. They are ideal for producing products such as acrylate esters, phthalate esters, and polyester polymers. The reasons Tyzor® titanates are better alternatives to sulfuric acid, sulfonic acid, and other metal-based Lewis acids are listed below.
-
They are a nontoxic organometallic chemistry.
-
They can be easily removed from esterification and transesterification products.
-
Their residue from catalysis is nontoxic.
-
They promote safer overall synthesis reaction conditions.
-
Higher ester conversion rates can be achieved in fewer cycles than strong acids.
-
Higher ester yields are achieved from acids and alcohols.
-
Lower esterification and transesterification byproduct formation.
-
Lower color and odor formation.
Selecting the proper Tyzor® catalyst for your esterification reaction is critical for maximizing the efficient production of esters and other chemical compounds. Contact us today to discuss your esterification or transesterification reaction with one of our product experts and find the grade of Tyzor® best suited for your application.
Are you in the correct region?
We’ve detected that you are located in a different region than the region selected on the website. Would you like to change your region?
Current Region: English - Canada