Select Your Region
Ethylene Glycol vs. Propylene Glycol

Thermal Fluid Chemistries Explained
Dow Chemical provides two basic types of low-temperature thermal fluids with their DOWTHERM™ (ethylene glycol based) and DOWFROST™ (propylene glycol based) portfolio. Both chemistries are well established as effective solutions for temperature control, but how do they differ and which solution is the right fluid for your requirement? This article aims to simplify & answer those questions to ensure proper thermal fluid seleciton.
Thermal Fluid Expertise - DOWFROST™ & DOWTHERM™
DOWFROST™ and DOWTHERM™ are the go-to solution for low-temperature heat transfer requirements, with more than 30 years’ worth of demonstrated success in HVAC systems, food processing facilities, and other equipment exposed to moderate temperatures (<350°F). There are slight differences in the chemical make-up of these products, but separate and individual handling & operating procedures are not specifically required.
There are three major factors that you should evaluate when deciding between a propylene glycol and an ethylene glycol solution:

Relative Toxicity
Ethylene Glycol (EG) is rated as moderately toxic if ingested, with a lethal dose being approximately 100mL or 3 ounces. In industrial settings where workers may be exposed to EG by inhalation, skin or eye contact – health concerns are relatively minor.
Propylene Glycol (PG) exhibits low oral toxicity and has seen extended term use in food & pharma products. Industrial exposure is generally not a health concern. Although these fluids differ slightly, it is recommended to treat both products with equal precautions, safe handling & processes.
Propylene Glycol (PG) exhibits low oral toxicity and has seen extended term use in food & pharma products. Industrial exposure is generally not a health concern. Although these fluids differ slightly, it is recommended to treat both products with equal precautions, safe handling & processes.

Regulatory Impact
Ethylene and Propylene glycol do not appear on the EPA list of “hazardous substances”. However, DOWTHERM™ is subject to reporting requirements for the Superfund Amendments & Reauthorization Act (SARA) and the Comprehensive Environmental Response, Compensation & Liability Act (CERCLA) due to its acute oral toxicity and air pollutant potential.
PG is considered GRAS (generally recognized as safe) by the FDA under 21 CFR 187.1666 and can be used in a wide array of food, beverage & pharmaceutical equipment.
PG is considered GRAS (generally recognized as safe) by the FDA under 21 CFR 187.1666 and can be used in a wide array of food, beverage & pharmaceutical equipment.

Heat Transfer Efficiency
PG is more viscous than EG, especially at very low temperatures. Resulting in potential loss in efficiency for cooling applications.
In order to best determine heat transfer efficiency in an air-cooled heat exchanger, you can use the following equation;

When used at a 50% dilution in water (Table 1 below), a measureable difference in efficiency is seen between EG (4.7% Loss) and PG (11.7% loss) due to the heavier viscosity in the PG fluid.
In order to best determine heat transfer efficiency in an air-cooled heat exchanger, you can use the following equation;

When used at a 50% dilution in water (Table 1 below), a measureable difference in efficiency is seen between EG (4.7% Loss) and PG (11.7% loss) due to the heavier viscosity in the PG fluid.
Table 1: Heat Transfer Comparison (50% EG) with (50% PG)
| Temp °F | Re (EG)
|
Re (PG)
|
hi (EG)
|
hi (PG)
|
A†
|
U (EG)
|
U (PG)
|
B††
|
|---|---|---|---|---|---|---|---|---|
| 170 | 54,612
|
45,564
|
968
|
855
|
11.7
|
18.69
|
18.64
|
99.7
|
| 120 | 31,554
|
23,648
|
739
|
628
|
15.0
|
18.58
|
18.49
|
99.5
|
| 70 | 15,318
|
8,983
|
508
|
356*
|
29.9
|
18.36
|
18.09
|
98.5
|
| 25 | 6,152
|
2,478
|
275*
|
121*
|
56.0
|
17.82
|
16.46
|
92.4
|
| 0 | 3,054
|
947
|
151*
|
--*
|
--*
|
16.92
|
--**
|
--
|
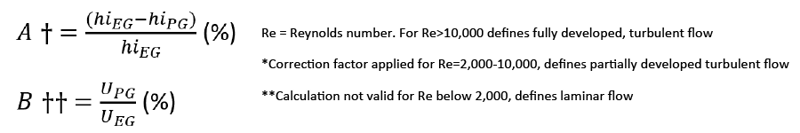
Table 2: Physical Properties Comparison EG and PG
Ethylene Glycol (100%)
|
Propylene Glycol (100%)
|
Ethylene Glycol (50% Solution)
|
Propylene Glycol (50% Solution)
|
|
|---|---|---|---|---|
| Molecular Weight | 62.1
|
76.1
|
--
|
--
|
| Freeze Point | 8°F (-13°C)
|
-71°F (-58°C) Tg
|
-34°F (-36°C)
|
-29°F (-33°C)
|
| Specific Gravity | 1.110
|
1.033
|
1.082
|
1.050
|
| Density (lb/ft^3) | 69.12
|
64.32
|
67.05
|
65.14
|
| Flash Point | 240°F (115°C)
|
220°F (104°C)
|
None
|
None
|
| Boiling Point | 387°F (198°C)
|
369°F (188°C)
|
225°F (108°C)
|
222°F (105°C)
|
Physical Properties Explained

Viscosity
PG is more viscous than EG, this difference is especially noticeable at lower temperatures where viscosity increases. When operating at ~170°F the viscosity difference between EG and PG is about 10% and generally unnoticeable. At colder temperatures and start up conditions, PG will require significantly more energy.

Freeze Point
Propylene Glycol has a higher molecular weight than EG and is less efficient in lowering the freezing point of water. Approximately 2% higher concentration of PG is required to give equivalent protection from freeze as an EG based solution.
Specific Gravity
The specific gravity of propylene glycol is very similar to that of water, and as a result it is impossible to identify specific gravity of PG solutions the same way as can be done for EG solutions. At a high level, one gallon of PG will weigh less than EG.
Solubility Parameter
PG has a greater affinity for oil-like materials than EG, this is reflected in the lower solubility parameter. PG acts slightly better as a solvent than EG and can have a greater effect on some plastics/elastomers at higher temperatures.Are you in the correct region?
We’ve detected that you are located in a different region than the region selected on the website. Would you like to change your region?
Current Region: English - United States