Select Your Region
Chevron Phillips Sulfide Agents

Optimize Your Refinery’s Hydrodesulfurization Activity
Dimethyl Sulfide (DMS) and TBPS 454 from Chevron Phillips Chemical are sulfiding agents that are used by refiners to transform metal oxide species to a metallic sulfide crystalline phase in situ for hydrotreating catalysts.
Sulfiding 101
Sulfur is a naturally occurring substance in petroleum fuels. However, in an effort to reduce harmful SOx and NOx emissions, the United States E.P.A (Environmental Protection Agency) began mandating the use of low-sulfur fuel. The main process refineries use to remove sulfur compounds from gasoline, diesel and jet fuel is called hydrotreatment or hydrodesulfurization.
Catalytic hydrotreating is a catalytic refining process widely used to remove sulfur, unsaturated hydrocarbons, and nitrogen from petroleum products such as naphtha, gasoline, diesel fuel*, kerosene, and fuel oil.
It is also referred to as hydroprocessing or hydrodesulfurization, commonly occurs at multiple locations in a refinery.
Hydrodesulfurization processes typically include facilities for the capture and removal of the resulting hydrogen sulfide (H2S) gas, which is subsequently converted into elemental sulfur or sulfuric acid, as byproducts.
The processes are used to ensure that petroleum products meet performance specifications and involve a number of chemical conversions.
During hydrotreatment, catalysts inside the reactor, typically CoMo (Cobalt Molybdenum) or NiMo (Nickel Molybdenum) convert unwanted sulfur and nitrogen compounds into H2S (hydrogen sulfide) and NH3, which is then removed in downstream processing. In order to activate catalytic activity, the hydrotreating catalysts go through a process called "sulfiding" or "presulfiding" where metal oxides are reacted with H2S in the presence of hydrogen. Sulfiding agents are used to readily decompose sulfur compounds into the H2S required to complete the sulfiding reactions. Two of the most common sulfiding agents are TBPS 454 and DMS. Below are the advantages of each grade:
TBPS 454 is a sulfiding agent with 54% sulfur content, used to transform metal oxide species to a metallic sulfide crystalline phase in situ for hydrotreating catalysts. For in situ sulfiding, the reaction is performed inside the process unit for complete control and to achieve maximum catalyst activity and safe handling. Below are the advantages:
- TBPS 454 is not classified as a flammable material and has a low toxicity rating.
- TBPS 454 has a diesel-like odor, a more friendly and common smell for refineries.
- TBPS 454 starts to convert to H2S at the lowest temperature compared to other sulfiding agents, lowering the risk of reduction of metals and potentially lessening the time of the sulfiding operation.
- The hydrocarbon byproduct during a sulfiding operation for TBPS 454 is isobutane, which does not have the potential to dilute hydrogen in the recycle gas during a sulfiding operation.
Dimethyl Sulfide (DMS) is a presulfiding agent with 51% sulfur content for steam cracking, the process that turns hydrocarbons into ethylene and propylene, which is used for olefin-based products. DMS is added to the feedstock to reduce coke and carbon monoxide formation during the high temperature thermal cracking of hydrocarbons in the tubular reactors. Below are the advantages:
- DMS directly and completely decomposes to H2S at 1470°F (although some trace CS2 may remain), suitable for furnace operating temperatures.
- DMS usually requires only one injection point in the charge gas as DMS will disperse fairly well throughout the furnace system.
Dimethyl Disulfide (DMDS)
Dimethyl Disulfide (DMDS) is a commercially used compound as a sulfur source in the steam cracking of hydrocarbons. It has a sulfur content of 65% and it is used as a solvent in various industrial processes, including petroleum refining, chemical sysnthesis and pharmaceutical manufacturing. In chemical synthesis as a precursor to other chemicals in organic synthesis. Also, as insecticide and fungicide, acting by disrupting the respiratory system of pests and pathogens.
- DMDS can be synthesized through several methods, including the reaction of methanol with hydrogen sulfide in the presence of a catalyst or the reaction of methanol with sulfur.
- DMDS has a lower ozone- depleting potential and reduced environmental impact, therefore, it contributes to a more sustainable farming practice
Sulfiding Agent Properties
| Property | DMDS
|
TBPS 454
|
DMS
|
|---|---|---|---|
| Sulfur %(wt) | 68
|
54
|
52
|
| Density (lbs/gal) | 8.9
|
9.0
|
7.11
|
| Freezing Point (°F) | -121
|
-54
|
-145
|
| Boiling Point (°F) | 229
|
N/A
|
99
|
| Flash Point | 59
|
217
|
-36
|
| Vapor Pressure at 70°F (PSI) | 0.45
|
<0.1
|
8.1
|
| Decomposition Temperature* (°F) | 392
|
320
|
482
|
| Auto Ignition Temperature (°F) | 575
|
437
|
403
|
| Viscosity at 70(°F) (mPa.s) | 0.62
|
12.8
|
.285
|
*In the presence of H2 and Catalysts
TBPS Typical Sulfiding Procedure
The sulfiding process should be carefully monitored and controlled to achieve the optimum sulfur activity. Below is a typical sulfiding procedure for TBPS 454. When decomposed, TBPS 454 can form elemental sulfur that forms solid polymers when reacted with olefins. The solid polymers can form residues on the catalyst bed and in the piping. There is a much higher risk of this happening at temperatures below 500 F. To decrease the likelihood of polymer formation, the injection point should be as near as possible to the catalyst bed.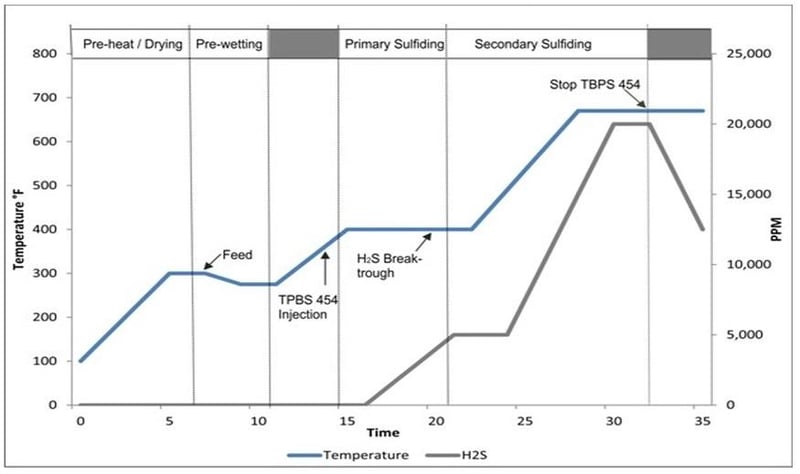
Are you in the correct region?
We’ve detected that you are located in a different region than the region selected on the website. Would you like to change your region?
Current Region: English - United States