Select Your Region
Pigment Wetting Agents & Dispersants
An Introduction to Pigments
Pigments are solid, insoluble powders that provide color to various materials, including paints, coatings, and inks. They are commercially available in a wide range of colors and chemistries. While there are many different options, they all have something in common: they must be properly dispersed for optimal performance and appearance in paint, coating, and ink formulations.What is Pigment Dispersion?
Pigment dispersion is the process of suspending insoluble pigments in a liquid medium, typically water or an organic solvent. Pigments have a strong tendency to agglomerate and are quite difficult to separate. Therefore, dispersing pigments is necessary to improve color strength, achieve even distribution of color in formulations, and prevent defects in film coverage and damage to dispensing equipment. Traditionally, pigment dispersion is comprised of three main steps: wetting, deagglomeration, and stabilization. These three steps are outlined in further detail below.

Pigment Wetting
When solid pigments are first added to the liquid medium, they are typically agglomerated together, with air entrapped on and within the pigment agglomeration. For proper wetting to occur, the air entrapped in the pigment must first be replaced with the liquid medium. Using an additive called a surfactant or wetting agent reduces surface tension and enables the liquid to achieve complete wetting of the pigment more easily.
Deagglomeration
Once complete wetting of the pigment particles is achieved, the large agglomerations of pigment must be broken up to reduce the particle size. This is commonly achieved through mechanical shear via a grinding mill. The quality of the grind is typically verified by color strength, visual assessment, and particle size quantification using grind gauges in accordance with test methods such as ASTM D1210. Reducing the surface tension in the liquid medium by the pigment wetting agent in the previous step allows for faster deagglomeration and a more homogeneous particle size in the dispersion.
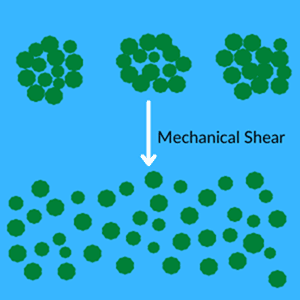
Stabilization
Once dispersed, the deagglomerated pigment particles must be stabilized to prevent reagglomeration and keep particles separated. In this step, pigment dispersion additives are added that adsorb onto the pigment particles and keep them separated. Stabilizing pigments in suspension can be achieved by either electrostatic or steric means. Once made, pigment dispersions can be validated for quality over time by viscosity analysis and light scattering equipment to determine the presence of agglomerates.
Electrostatic Dispersants
Electrostatic dispersant additives carry an ionic charge. They interact with the surface of the pigment particles to produce like-charged surfaces, which are typically cationic. These like charges on the surface of the pigment particles repel each other and keep the pigment particles separated in paint, coating, and ink formulations. Changing pH or adding ionic material can destabilize particles suspended with electrostatic dispersants.
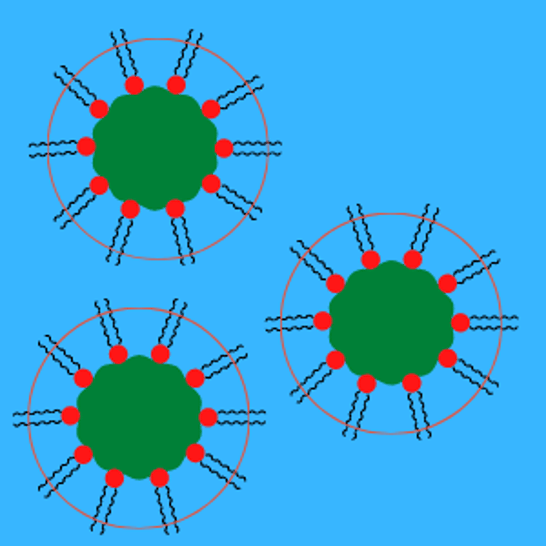
Steric Dispersants
Steric dispersant additives are molecules with an anchoring head group attracted to the pigment particles and tails attracted to the liquid phase of the dispersion. The head groups adsorb onto the surface of the pigments, and the tails orient themselves outward from the surface. The protruding tails repel each other and keep the pigment particles separated and suspended. Steric stabilization is incredibly stable at high salt concentrations and over a wide pH range.
Why are Pigment Wetting Agents & Dispersants Important?
In many cases, the pigment is the most expensive ingredient in an ink, paint, or coating formulation because of its cost and the high concentration it is used. Optimizing the wetting and dispersion of pigments ensures optimal appearance and physical properties in the formulation while using the least amount of pigment possible. Selecting the right wetting agent and dispersion additive for coating, ink, and paint formulations is critical for producing high-quality products with minimized production costs. .Surfactants as Wetting Agents and Dispersants
Surfactants, or surface-active agents, are additives that reduce surface tension at the interface between two substances. In the case of wetting and dispersing pigments, reducing the surface tension between the solid pigment particles and the liquid carrier allows for faster and more efficient wetting and deagglomeration. Additionally, surfactants can adsorb onto the surface of pigment particles and provide steric stabilization of the dispersion. Commercially, many types of nonionic surfactants and anionic surfactants are used today as wetting agents and dispersants in inks, paints, and coatings. These surfactants are compared below based on performance characteristics.
| Surfactant Type | Reduction of Surface Tension | Foam Tendency
|
Addition Rate (%)
|
|---|---|---|---|
| Alcohol Ethoxylate | Moderate
|
Moderate
|
1.00–3.00
|
| Sulfosuccinate | Good
|
High
|
1.00–3.00
|
| Fluorosurfactants | Excellent
|
Low
|
0.01–0.05
|
| Polyether Siloxane | Moderate
|
Moderate
|
1.00–3.00
|
| Acetylene Diol | Good
|
Low
|
0.10–1.00
|
Selecting the appropriate surfactant for pigment wetting and dispersion should be based on the liquid carrier (aqueous or organic solvent), the pigment, and the end-use application. ChemPoint offers various surfactant chemistries that can be used for pigment wetting and stabilization applications. These are listed below.
We are happy to provide formulators with samples, pricing, and technical assistance. Click below to discuss your project with one of our technical experts.
Are you in the correct region?
We’ve detected that you are located in a different region than the region selected on the website. Would you like to change your region?
Current Region: English - United States